[column width=”1/3″ place=”first” ] [headline margin=”no-margin” htype=”h4″ invertclass=”invertblock”]IBDoc® Kit[/headline]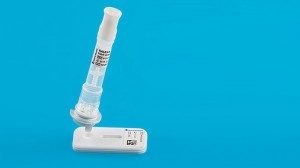
The CALEX® Valve is an easy to use device designed by BÜHLMANN which allows precise and quantitative stool extraction to be used for calprotectin home testing. It consists of the sampling pin, the body and a valve to release a precise amount of stool extract onto the test cassette. The body is pre-filled with extraction buffer and thus delivered ready to use.
[/column] [column width=”1/3″ place=”none”] [headline margin=”no-margin” htype=”h4″ invertclass=”invertblock”]CalApp®[/headline]![]()
The CalApp® turns your smartphone into a test cassette reader using state of the art image processing. First the CalApp® guides the user through the test procedures step by step with an intuitive tutorial. Once the test cassette is ready, the CalApp® scans the cassette and calculates an quantitative calprotectin concentration. The CalApp® is available for iOS and Android.
[/column] [column width=”1/3″ place=”last” ][headline margin=”no-margin” htype=”h4″ invertclass=”invertblock”]IBDoc® Portal[/headline]
The IBDoc® Portal serves as a platform for Health Care Professionals to register and manage patient information and to follow their test results over time. The Health Care Professionals are immediately notified about every new test result of one of their patients. The IBDoc® Portal is designed with the highest security standards to keep confidential information safe and serves as communication platform between patient and doctor.
[/column]
[headline margin=”margin” htype=”h3″ triangle=”yes”] Test Procedure [/headline][column width=”1/3″ place=”first” ] [headline margin=”no-margin” htype=”h4″]1 Prepare Test[/headline]

After downloading and starting the CalApp® the patient logs in with his or her IBDoc® account username and password and is ready to start the test procedure. The first step is to collect a stool sample with the easy to use stool collection paper sheet included in the IBDoc® Test Kit.[/column] [column width=”1/3″ place=”none”] [headline margin=”no-margin” htype=”h4″]2 Extract Sample[/headline]

After collection of the stool sample, the pin of the CALEX® Valve extraction device is dipped into the sample a few times with a twisting motion so that a predefined amount of stool sticks to the grooves of the CALEX® Valve sampling pin. Then the pin is placed back into the tube through the upper funnel.[/column] [column width=”1/3″ place=”last” ][headline margin=”no-margin” htype=”h4″]3 Load Test Cassette[/headline]
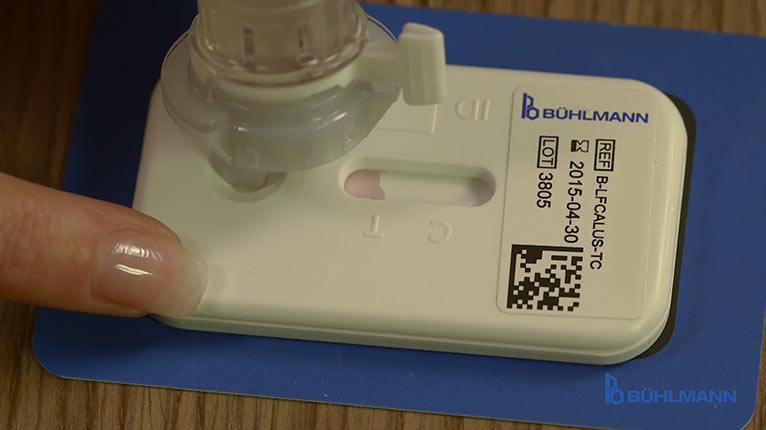
The CALEX® Valve tube is applied onto the cassette and a precise amount of extract is released by turning the throttle. After a few seconds, when the membrane strip of the cassette gets visibly soaked, the throttle is closed.[/column]
[column width=”1/3″ place=”first” ] [headline margin=”no-margin” htype=”h4″]4 Read Test Cassette[/headline]

To read the result the camera is placed above the cassette, the overlay is focused and a picture is automatically taken. The image is analysed through a sophisticated image processing and a quantitative result is calculated by the CalApp®.[/column] [column width=”1/3″ place=”none”] [headline margin=”no-margin” htype=”h4″]5 Save Test Result[/headline]
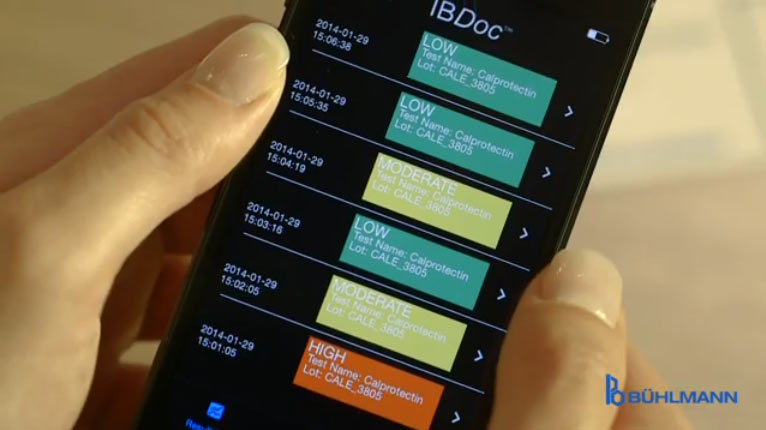
The result is classified as NORMAL, MODERATE, or HIGH, shown in a traffic light code and saved. All the results performed on one smartphone are saved locally and can be accessed at any time later on.[/column] [column width=”1/3″ place=”last” ][headline margin=”no-margin” htype=”h4″]6 Doctor Is Notified[/headline]

The result taken with the CalApp® is automatically transferred to the IBDoc® Portal and the patient’s doctor is immediately notified. Depending on the test result the doctor can communicate with the patients to advise on further therapeutic steps to be taken.[/column]
[column width=”1/3″ place=”first”] [headline margin=”no-margin” htype=”h4″]Reliable[/headline]
All components of the IBDoc® systems have been developed to measure up to the highest standards of the industry resulting in a robust and reliable test – the first of its kind. Quality is not just a statement, but part of BÜHLMANN’s daily commitment.
[/column] [column width=”1/3″ place=”none” ][headline margin=”no-margin” htype=”h4″]Easy to use[/headline]
The IBDoc® Test Kit provides an easy to use therapy monitoring home testing system for IBD Patients – empowering the patients, connecting them with their doctors and allowing them to manage the disease as a team.
[/column] [column width=”1/3″ place=”last” ] [headline margin=”no-margin” htype=”h4″]Secure[/headline]
BÜHLMANN takes great care that patient information is transmitted and stored in a secure way. The IBDoc® Portal works with online-banking grade data encryption and only a patient’s treating physician has access to medical information.
[/column]
[headline margin=”margin” htype=”h3″ triangle=”yes”] What are our customers saying? [/headline][column width=”3/3″ place=”first” ]
[column width=”3/3″ place=”l” ][testimonials][testimonial author=”Kathleen Sugrue” job=”IBD Nurse “]I do strongly believe it should be a standard of care that all patients have access to IBDoc®.[/testimonial][testimonial author=”Pearly Avery” job=”IBD Nurse “]IBDoc® integrates very well. It`s not more work at all. I think, if anything, it`s less work.[/testimonial][testimonial author=”Barb Harbers” job=”IBD Nurse Navigator”]IBDoc® has been an amazing addition to our care. We have definitely seen patients have a better understanding of their disease process and it has really improved patient management.[/testimonial][testimonial author=”Alana Carter from Ontario” job=”IBD Nurse”] I have had the opportunity to use IBDoc® within a community GI practice. It is a promising clinical test to obtain fecal calprotectin results with no delays or turnover. You are notified that the results are posted within the portal, that displays not only the results but a trending graph. Patients have expressed that it is easy to use, convenient and gives them a sense of participation in managing their disease. They are given the kit in the clinic, which eliminates a trip to the lab to pick up sample containers and the trip back to the lab to drop off samples for testing. In the clinic, I explain how to use IBDoc® kit at home and how their results will be interpreted. I feel that fecal calprotectin results are part of the patients overall clinical assessment to help guide decision making in regards to disease activity and treatment.[/testimonial][testimonial author=”M.K. from Vienna” job=”Student & Crohn’s Patient “] I think that IBDoc® is very useful in determining my Calprotectin value and it is incredibly easy to use. I think it is extremely handy! [/testimonial][testimonial author=”Sam Elfassy, MD FRCPC from Toronto” job=”Gastroenterologist”] IBDoc® has quickly become an invaluable tool for close follow-up in my patients with Ulcerative Colitis. My patients have been much happier with the quick results and the convenience of performing these tests at home compared to taking the samples to the lab. I have appreciated the benefit of being able to make changes to their management based on both symptoms and the objective measure of fecal calprotectin without the two-week turnaround I was accustomed to. The process alerts me to new results and the portal stores previous results so I can easily track my patients’ progress.[/testimonial][testimonial author=”iOS User” job=”via Apple App Store “]This app is a game changer! This app is a complete game changer for me. I’ve had Crohn’s disease for 28years. Having the ability to check disease activity via the IBDoc app in the comfort of your own home is a powerful tool indeed. The IBDoc app is intuitive and user friendly. I simply followed the in-App instructions, set the timer, and waited for 12 minutes. I then have the result!! Can’t rate it highly enough.[/testimonial][testimonial author=”I.S.” job=”via Google Play Store “]Excellent! Easy to use and very helpful.[/testimonial][/testimonials][/column]
[headline margin=”margin” htype=”h3″ triangle=”yes”] Marketing Material [/headline][column width=”3/3″ place=”first” ][/column]
[column width=”1/3″ place=”first” ][headline margin=”no-margin” htype=”h4″]IBDoc® Validated Smartphone Flyer[/headline] [/column]
[/column]
[column width=”1/3″ place=”none” ][headline margin=”no-margin” htype=”h4″]IBDoc® Information Brochure[/headline] [/column][column width=”1/3″ place=”last” ][headline margin=”no-margin” htype=”h4″]IBDoc® Clinical Publication Flyer[/headline]
[/column][column width=”1/3″ place=”last” ][headline margin=”no-margin” htype=”h4″]IBDoc® Clinical Publication Flyer[/headline] [/column]
[/column]
[headline margin=”margin” htype=”h3″ triangle=”yes”] About Calprotectin [/headline][column width=”3/3″ place=”first” ]
[headline margin=”low-margin” htype=”h4″]What is calprotectin?[/headline]
Calprotectin is a heterodimeric protein also known as MRP8/14 and is abundantly present in macrophages and neutrophil granulocytes; inflammatory cells of the immune system. Calprotectin is released from these cells at the site of active inflammation and therefore elevated calprotectin levels indicate an ongoing inflammation. Calprotectin can be measured in both blood and faecal samples.[headline margin=”low-margin” htype=”h4″]Why measuring faecal calprotectin?[/headline]
Measurement of faecal calprotectin is considered a reliable indicator of inflammation and numerous studies show that while faecal calprotectin concentrations are significantly elevated in patients with inflammatory bowel disease (IBD), patients suffering from non-organic bowel disease (IBS) do not have increased calprotectin levels. Such increased levels are shown to correlate well with both endoscopic and histological assessment of disease activity.
[headline margin=”low-margin” htype=”h4″]What is IBD?[/headline]
Around 2 million people in Europe suffer from IBD. Crohn’s Disease (CD) and Ulcerative Colitis (UC) are incurable serious chronic diseases of the intestinal tract. The symptoms are distressing, embarrassing and even debilitating. Once symptoms appear, they resemble those of other conditions of functional origin (IBS), which make it very difficult for doctors to correctly diagnose.
[headline margin=”low-margin” htype=”h4″]Why measuring faecal calprotectin in IBD Patients?[/headline]
The clinical course of most patients with IBD is marked by periods of remission with intermittent relapses characterized by increased intestinal inflammation. Numerous published studies have looked at faecal calprotectin levels in patients during the course of the disease. The results show that calprotectin is a good predictor of relapse in patients with IBD. The treatment aim in IBD patients is of course clinical remission. Because sustained clinical remission is associated with mucosal healing in the gut and reduced rates of hospitalisation, identifying patients likely to relapse enables their clinicians to adapt the patients treatment accordingly and to ease the relapse severity.
[/column]
[headline margin=”margin” htype=”h3″ triangle=”yes”] About BÜHLMANN[/headline][column width=”1/3″ place=”first” ] [/column][column width=”2/3″ place=”last” ] [headline margin=”notop-margin” htype=”h4″]BÜHLMANN Laboratories AG – A COMMITMENT TO DIAGNOSTICS[/headline]
[/column][column width=”2/3″ place=”last” ] [headline margin=”notop-margin” htype=”h4″]BÜHLMANN Laboratories AG – A COMMITMENT TO DIAGNOSTICS[/headline]
BÜHLMANN is a fully independent, medium sized and family owned Swiss company. Founded in 1976, BÜHLMANN today has local teams in Switzerland, France, Italy, Germany, Austria, Brasil, the USA and distributors worldwide.
BÜHLMANN has become the prime calprotectin company within few years, offering the broadest range with a fully automatable, high-throughput BÜHLMANN fCAL™ ELISA and the quantitative Quantum Blue® Calprotectin rapid tests. BÜHLMANN does not only stand for calprotectin, but a selection of unique, high-quality assays for clinical routine use such as the BÜHLMANN Flow CAST® product group for Basophil Activation Testing, and the BÜHLMANN GanglioCombi™ products, the only screening ELISAs for anti-ganglioside testing. As unique as our products are our distribution fields with leading suppliers in the field of molecular diagnostics.
The Quality Management System is ISO 9001:2008 and 13485:2003 certified and the products are manufactured according to GMP standards.
www.buhlmannlabs.ch[/column]
*IBDoc® is not available in the US.